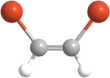
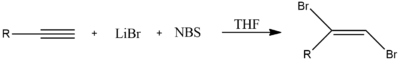1,2-Dibromoethylene
1,2-Dibromoethylene, also known as 1,2-dibromoethene and acetylene dibromide, is a dihalogenated unsaturated compound with one bromine on each of the two carbon atoms. There are two isomers of this compound, cis and trans. Both isomers are colorless liquids.
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1,2-Dibromoethene | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
| ECHA InfoCard | 100.007.953 | ||
| EC Number |
| ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C2H2Br2 | |||
| Molar mass | 185.846 g·mol−1 | ||
| Appearance | colorless liquid | ||
| Density | 2.246 g/cm3 | ||
| Boiling point | 110 °C (230 °F; 383 K) | ||
| Hazards | |||
| Main hazards | Toxic | ||
| GHS pictograms |    | ||
| GHS Signal word | Danger | ||
GHS hazard statements |
H301, H314, H315, H319, H335 | ||
| P260, P261, P264, P270, P271, P280, P301+310, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P310, P312, P321, P330, P332+313, P337+313, P362, P363, P403+233, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Synthesis
(E)-1,2-Dibromoethene can be synthesized by halogenation of acetylene with bromine Br2.[1] In order to prevent the formation tetrahalogenated compounds, acetylene is used in excess, with Br2 as the limiting reagent.
Alternately, halogenation of this kind could also be achieved through the use of two equivalents of N-bromosuccinimide and lithium bromide (LiBr). N-Bromosuccinimide provides Br+ as an electrophile, which is followed by Br− from LiBr.[2]
gollark: That is a terrible example.
gollark: 🇦 🇳 🇾 🇼 🇦 🇾 ,🇪 🇽 🇵 🇱 🇦 🇮 🇳 🇹 🇭 🇪 🇴 🇴 🇵 🇮 🇫 🇾 🇴 🇺 🇲 🇺 🇸 🇹 ...
gollark: 🇮 🇼 🇴 🇷 🇰 🇪 🇩 🇭 🇦 🇷 🇩 🇴 🇳 🇲 🇴 🇩 🇮 🇫 🇾 🇮 🇳 🇬 @🇺 🇲 🇳 🇮 🇰 🇴 🇸 '🇨 🇴 🇩 🇪 🇸 🇱 🇮 🇬 🇭 🇹 🇱 🇾 .
gollark: 🇳 🇪 🇻 🇪 🇷
gollark: 🇮 🇲 🇪 🇦 🇳 ,🇹 🇭 🇪 🇾 🇨 🇦 🇳
References
- "Chapter 9: Addition Reactions of Alkynes". Organic Chemistry 4e Carey. McGraw-Hill. Archived from the original on April 12, 2016. Retrieved 9 June 2017.
- Shao, L.-X.; Shi, M. (2006). "N-Bromosuccinimide and Lithium Bromide: An Efficient Combination for the Dibromination of Carbon–Carbon Unsaturated Bonds" (PDF). Synlett. 2006 (8): 1269–1271. doi:10.1055/s-2006-941558.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.
-1%2C2-Dibromoethene.png)
-1%2C2-Dibromoethene.png)
.jpg)